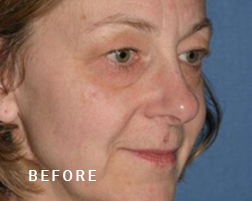
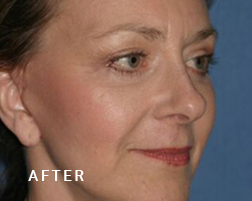
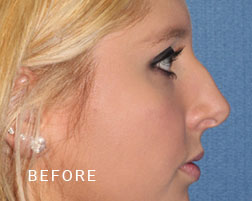
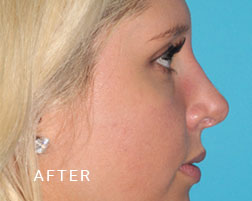
Breast Implant Illness (BII)
Offered at our convenient location in Scottsdale, Arizona
Breast Implant Illness is a term used to describe the symptoms that many women experience after undergoing a breast augmentation with synthetic implants. While symptoms vary from patient to patient, they commonly include muscle and joint pain, difficulty concentrating, impaired memory, depression and anxiety, and weight fluctuations. (1) Frustratingly, as the Food and Drug Administration reports, there has been no definitive cause identified linking implants to these symptoms, and breast implant illness is not considered a formal medical diagnosis.
Dr. Andrew Wolin puts his patients’ safety first, and although most women enjoy their breast implants in perfect health, he is dedicated to educating his patients about any risks associated with the services that he provides. If you’re heartbroken at the thought of losing your implants or perplexed by the onset of inexplicable symptoms, you have options.
Schedule a personal consultation today to learn more and make an informed decision. It’s your body and your health. Call (480) 945-8440 to reach our Scottsdale office and make an appointment.
Contents
About Breast Implant Illness
Breast implant illness (BII) has become a highly contested topic in the world of cosmetic surgery and medical device regulation. Although the symptoms are widely documented, researchers have had little success in tying these devices to the illness. Part of the difficulty lies in the wide variety of generalized symptoms that women attribute to BII. Fatigue, anxiety, inflammation, and other symptoms affect the entire body and are difficult for doctors to attribute to any one cause.
Another challenge is that there is no documented average for the onset of these symptoms after the implant is placed. Some women start to experience symptoms only a year or two after their surgery, while others feel fine until a decade or so later. However, women know what they’re feeling, and many of them are certain that their breast implants are to blame.
Women report symptoms of BII that include:
- “Brain fog” or trouble with memory and concentration
- Hair loss
- Fatigue
- Chest pain
- Trouble sleeping
- Irritable bowel syndrome
- Headaches
- Body-wide chronic pain
- Autoimmune diseases, such as lupus and fibromyalgia
BIA-ALCL
While BII isn’t recognized formally, another serious illness linked to breast implants is: Breast Implant Associated Anaplastic Large Cell Lymphoma (BIA-ALCL). This rare but dangerous cancer of the lymphatic system begins in the capsule of scar tissue that forms naturally around the synthetic breast implant. Dr. Wolin urges any woman that has undergone breast implants to be vigilant about their breast health and seek medical attention for any symptoms that could be a sign of this cancer.
Debated Devices
Did you know that from 1992-2006 there was a moratorium on silicone breast implants? As the New York Times reported when the FDA lifted this ban, in the 1970s and 1980s complaints about faulty devices and the side effects that they inflicted proliferated. Ruptured devices caused women to develop cancer in the most extreme cases, as well as autoimmune diseases. The makers of implants pulled their products from the market in 1992, and synthetic silicone implants were reserved only for women undergoing breast reconstructions or those enrolled in clinical studies. In fact, there were so many lawsuits that the behemoth Dow Corning was forced to file for bankruptcy to halt litigations. (3) However, after numerous studies (many of which were funded by the manufacturers of breast implants) the FDA declared that breast implants were safe for use in women over the age of 22, and in 2006, the moratorium was lifted.
Conflicting Studies
Nevertheless, research continues in order to better understand the effect this popular procedure has on the body. A recent study in the Netherlands examined tissue samples from 389 women with silicone breast implants. The samples were taken while the women were undergoing breast implant removal and included the capsules of tissue that had formed around their implants as well as the neighboring lymph nodes. When the tissues were tested, silicone was found in 98.8% of the samples, indicating silicone leakage had occurred, even though the implant was intact. Even more alarming, in 86.6% of these women, the silicone had migrated outside of the capsule. The presence of silicone in the lymph nodes suggests that breast implants could in fact be affecting the immune system, which may account for some of the symptoms associated with breast implant illness.
Silicone in tissues is associated with inflammatory reactions, as the body seeks to repair the perceived injury after detecting this toxin. (1) Unfortunately, research on this subject is scarce. However, another study of 100 women having capsules and implants removed in the United States revealed more troubling data. When tested upon removal, samples showed that the implants had been colonized by bacteria, including Staphylococcus, in nearly half of the cases. (2)
Rethinking Your Implants?
If you’re experiencing symptoms that you fear may be related to your breast implants, it’s important to seek medical attention to rule out the possibility of other underlying illnesses. But if you’ve decided that a bigger bustline isn’t worth the risk, there are procedures that can offer relief and still allow you to enjoy enhanced curves safely. Dr. Wolin provides a full array of breast procedures that can let you safely retain your feminine contours and enjoy peace of mind.
Breast Implant Exchange
Breast implants are not intended to remain in place for more than 10 years. As they remain in the breast cavity, the body responds by creating a layer of scar tissue around the implant. As this “capsule” of tissue continues to develop, it can put pressure on the implant leading to the painful condition known as capsular contracture. Aging breast implants are also more prone to rupture, which can affect the shape, feel, and health of the breast. By removing the capsule of tissue along with the implant, these complications can be avoided. And, since today’s technology has allowed for the development of safer, more durable breast implants, replacing your current implants could be an important decision. A breast implant exchange allows you to upgrade to a safer model, resolve symptoms, avoid complications, and gives you the opportunity to select a new size or shape.
Breast Implant Revision with Fat Transfer
If you enjoyed your breast implants but are experiencing symptoms or just want to eliminate any possibility of further complications, it may be time to start a new chapter. Breast implant revision will remove the implants and replace them with a fat graft taken from your own body. This procedure is truly a win win. Dr. Wolin uses liposuction to remove healthy, living (albeit unwanted) fat tissue from some of your most frustrating problem areas. Imagine a flatter abdomen, lithe thighs, or finally being rid of those pesky love handles! The fat is then purified and used to fill the breast cavity for a natural look and feel, and long-term volume that is all your own. There’s no risk of rejection, adverse reactions, or the complications associated with synthetic implants: just lush fullness that is 100% you.
A Safe Way Out
Removing the breast implant is the best way to resolve symptoms or risk of Breast Implant Illness. Although it was previously thought that the implant and the capsule removal would be best carried out as a combined procedure (composite capsulectomy) it has been found that it does not matter if the capsule is removed separately from the implant. As long as the capsular tissue is removed, the offending agent will be adequately eradicated. Dr. Wolin always performs all procedures to satisfy best practices in the best interest of his patients. He will utilize the technique that provides the best, most efficient, and healthiest outcome. As a trained scientist as well as aesthetic surgeon, he strives to always deliver surgical and non-surgical care based on sound scientific and clinical principles.
Personal Consultation
During your personal consultation with Dr. Wolin, he’ll review the details of your implant surgery and allow you the space to voice your concerns. You can expect a private, compassionate environment that will give you the information and resources you need to make a proactive decision about your breast implants. Safety comes first at our practice, which is why Dr. Wolin offers twice-yearly complimentary ultrasounds to ensure that his breast augmentation patients are in good health and able to continue enjoying their results. If additional diagnostic measures are required, he’ll review the results with you to make sure that you have a full understanding of your circumstances, and can make a confident decision as to how to proceed.
Moving Forward
Although the thought of undergoing additional surgery may be discouraging, Dr. Wolin and his staff ensure that all of their patients have the best possible experience, and the support they need to make this important decision.
Although there’s much we don’t know about BII, data shows that breast implant explantation (removal) has extremely high satisfaction rates. And, in the hands of a skilled and compassionate surgeon, you won’t have to compromise your aesthetic goals for safety. To get started, schedule a personal consultation or call (480) 945-8440 to reach our Scottsdale office. We’re ready to help.
References
- Dijkman HBPM, Slaats I, Bult P. Assessment of Silicone Particle Migration Among Women Undergoing Removal or Revision of Silicone Breast Implants in the Netherlands. JAMA Network Open. 2021;4(9):e2125381. doi:10.1001/jamanetworkopen.2021.25381
- Katsnelson JY, Spaniol JR, Buinewicz JC, Ramsey FV, Buinewicz BR. Outcomes of Implant Removal and Capsulectomy for Breast Implant Illness in 248 Patients. Plastic and Reconstructive Surgery Global Open. 2021;9(9):e3813. doi:10.1097/GOX.0000000000003813
- Saul S. F.D.A. Will Allow Breast Implants Made of Silicone. The New York Times. https://www.nytimes.com/2006/11/18/washington/18breast.html. Published November 18, 2006. Accessed October 19, 2021.